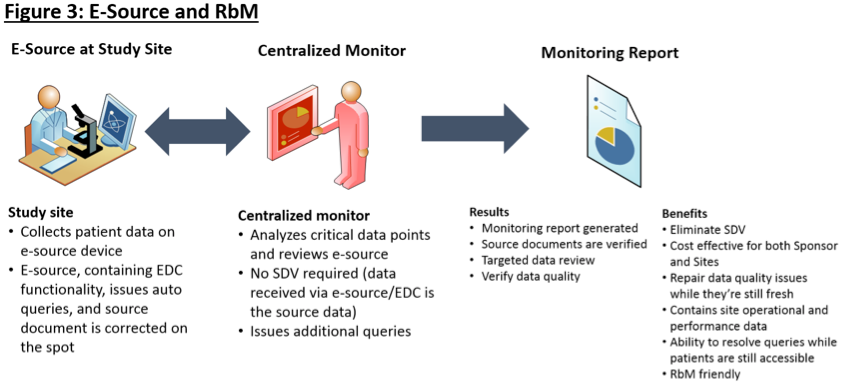
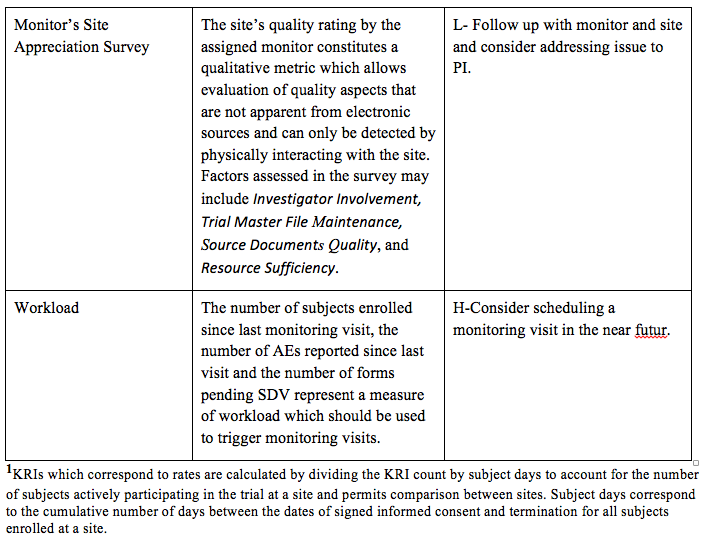
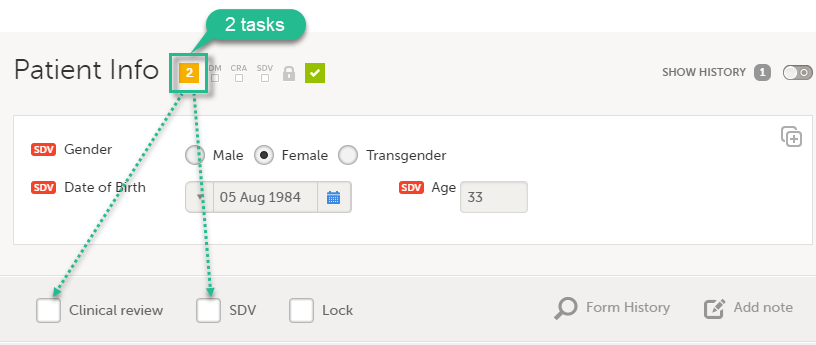
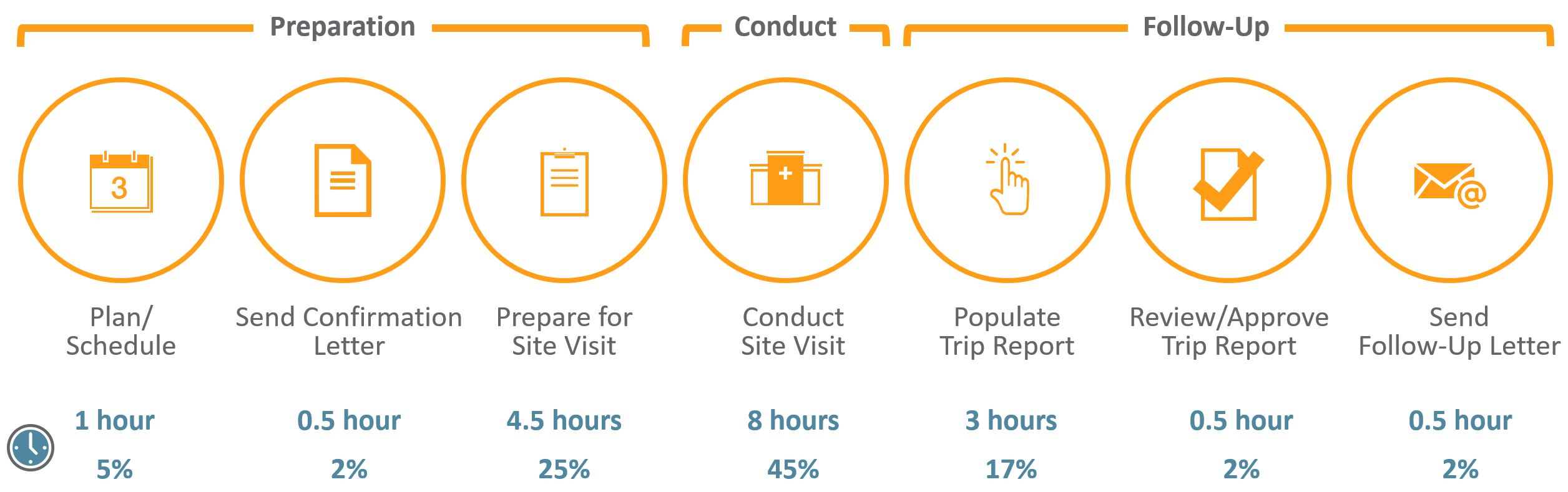
Assessing data quality and the variability of source data verification auditing methods in clinical research settings - ScienceDirect

Data cleaning and monitoring flow: Extended risk-based monitoring (RBM)... | Download Scientific Diagram

The Role of Source Data Verification (SDV) and Source Data Review (SDR) in Driving Clinical Trial Data Quality - Medidata Solutions

PDF) Extended Risk-Based Monitoring Model, On-Demand Query-Driven Source Data Verification, and Their Economic Impact on Clinical Trial Operations