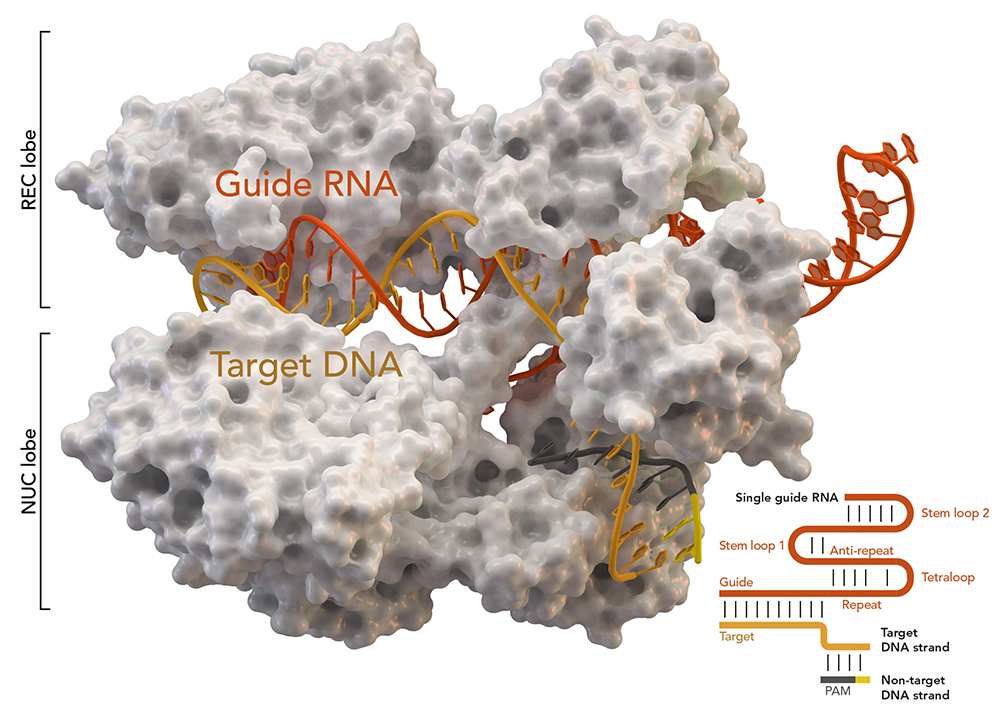
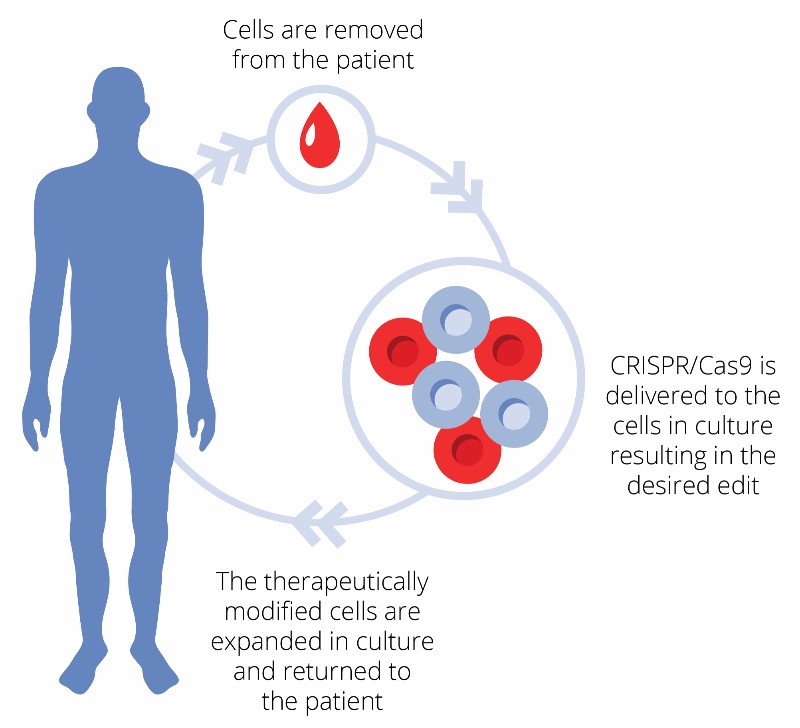
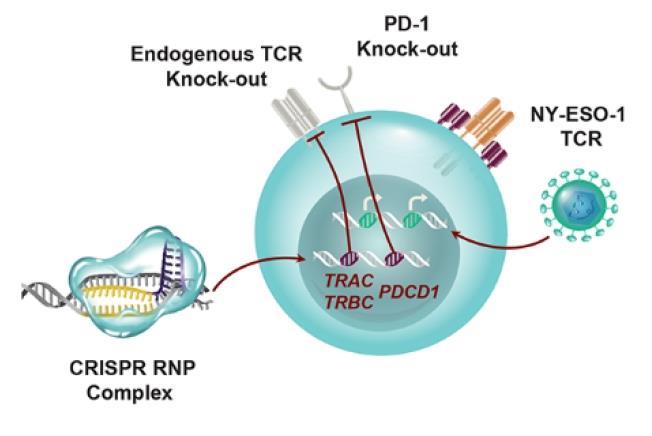
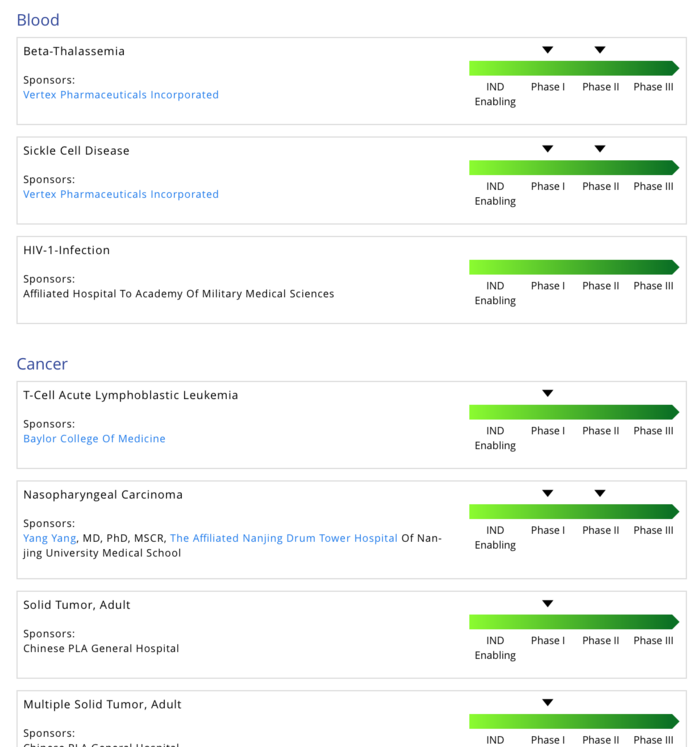
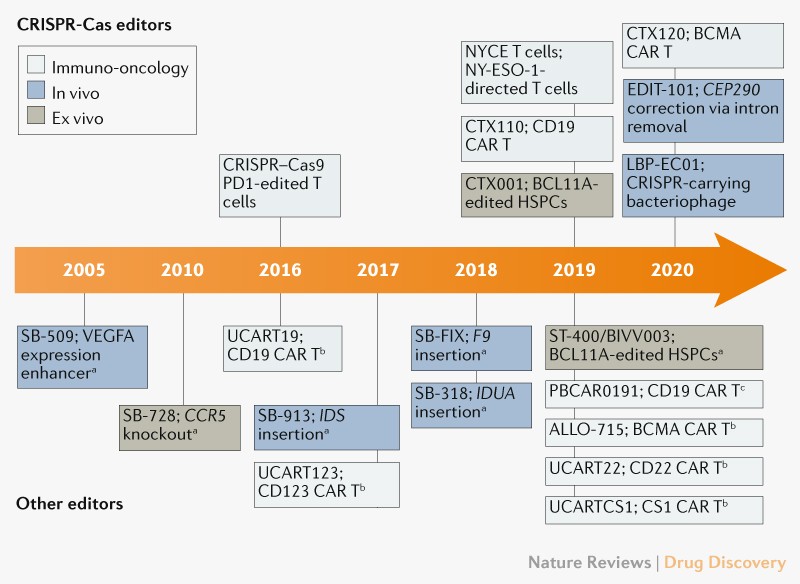
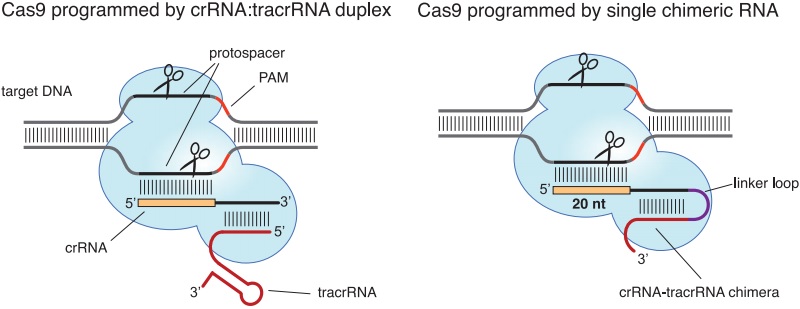
First Human Clinical Trial Finds CRISPR Gene-edited Cells Safe and Durable for Cancer Therapy | Technology Networks

The first human trial of CRISPR-based cell therapy clears safety concerns as new treatment for late-stage lung cancer | Signal Transduction and Targeted Therapy

Intellia's early CRISPR trial data validate a drug pipeline and the gene-editing field - MedCity News

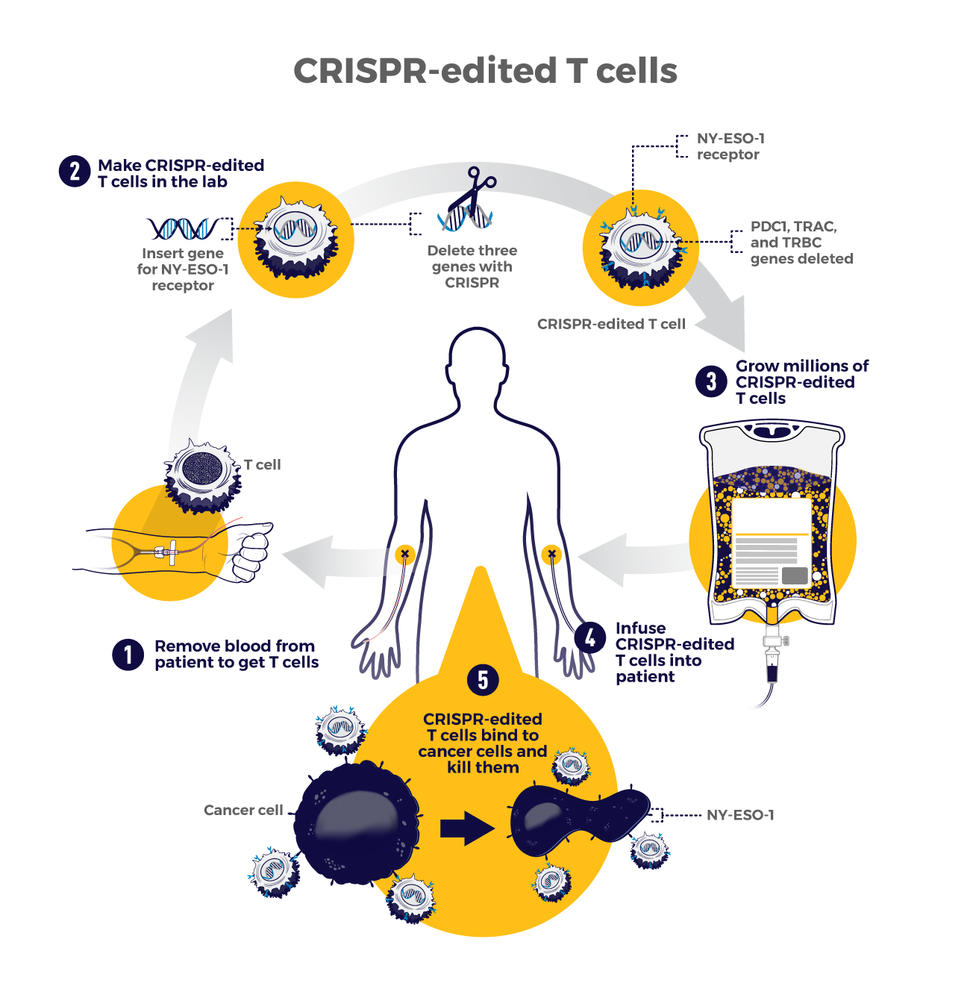
CRISPR-Edited Immune Cells Can Survive and Thrive After Infusion into Cancer Patients - Penn Medicine