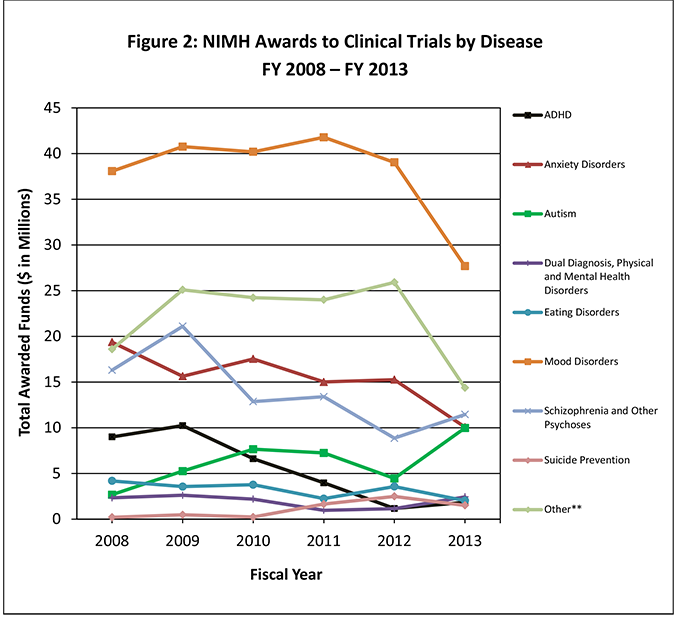
Comparative efficacy and tolerability of medications for attention-deficit hyperactivity disorder in children, adolescents, and adults: a systematic review and network meta-analysis - The Lancet Psychiatry

Vitamin-mineral treatment of attention-deficit hyperactivity disorder in adults: double-blind randomized placebo-controlled trial – Micronutrients Research
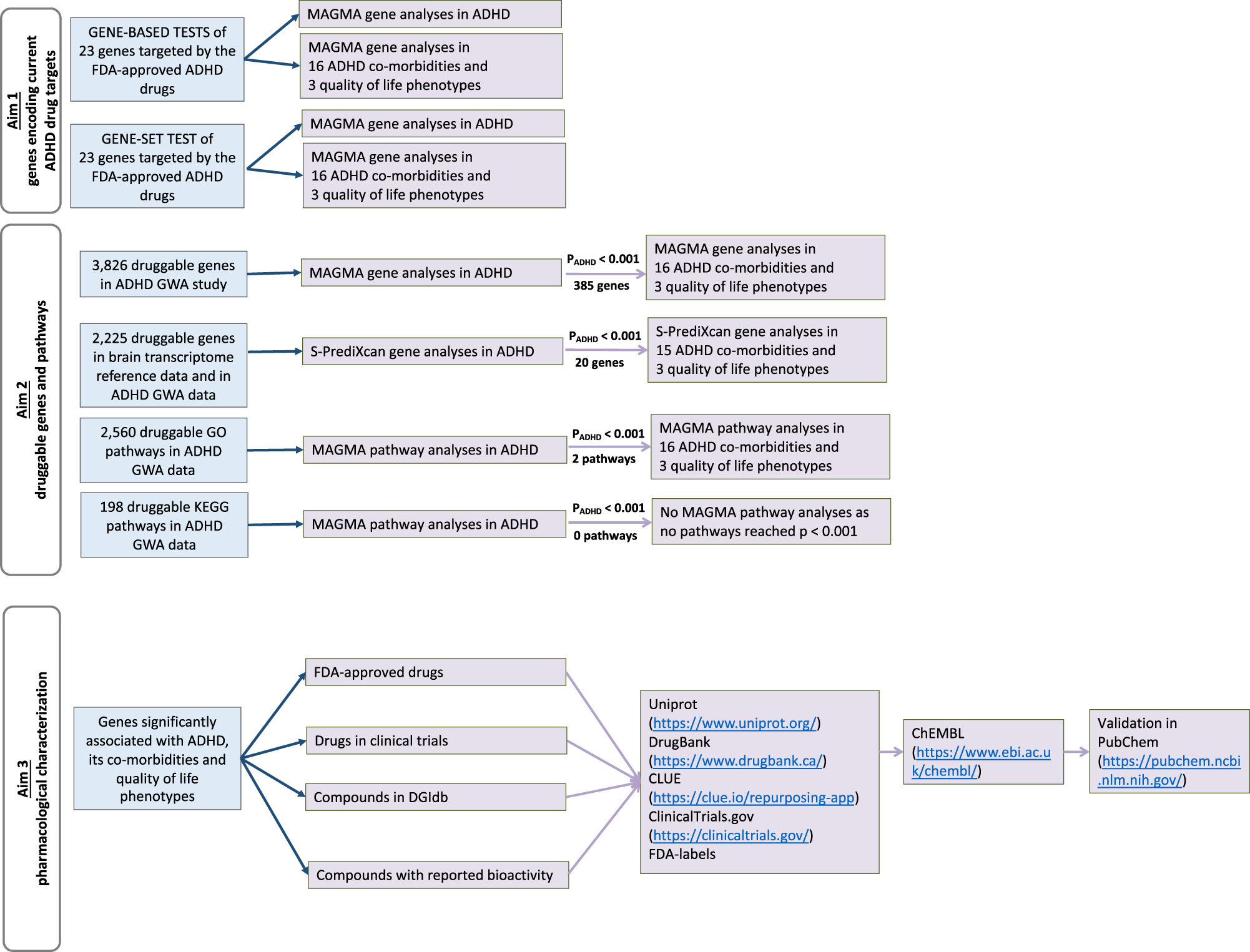
Druggable genome in attention deficit/hyperactivity disorder and its co-morbid conditions. New avenues for treatment | Molecular Psychiatry
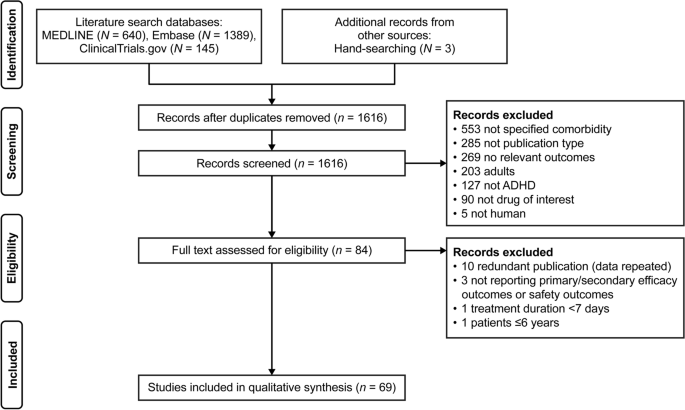
Efficacy and Safety of Medication for Attention-Deficit Hyperactivity Disorder in Children and Adolescents with Common Comorbidities: A Systematic Review | SpringerLink
PLOS ONE: Gastrointestinal adverse events during methylphenidate treatment of children and adolescents with attention deficit hyperactivity disorder: A systematic review with meta-analysis and Trial Sequential Analysis of randomised clinical trials

Clinical trials were missing from regulatory documents of extended-release methylphenidate for ADHD in adults: a case study of public documents - ScienceDirect

Introduction. Adult ADHD in the Real World: From Clinical Trials to Clinical Practice. - ppt download